This web page was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison.
What is Glucose-6-Phosphate Dehydrogenase (G6PD)?
|
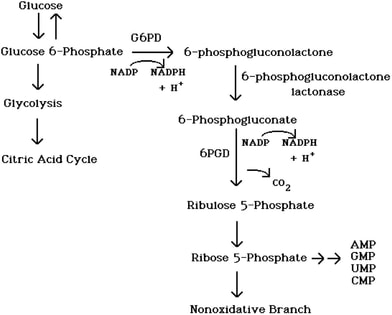
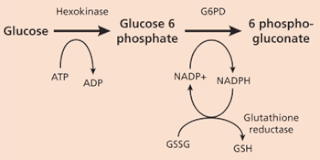
Glucose-6-phosphate dehydrogenase (G6PD) is a housekeeping enzyme, meaning it is expressed in all cells and required for the maintenance of cell function [1]. The G6PD enzyme is a part of the Pentose Phosphate Pathway. G6PD produces reduced Nicotinamid adenine dinucleotide phosphate (NADPH). NADPH protects cells from reactive oxygen species. NADPH is crucial in red blood cells, because when reactive oxygen species build-up, oxidative stress causes hemolysis (bursting of red blood cells). In red blood cells, the Pentose Phosphate Pathway is the only source of NADPH. Therefore, without functional G6PD enzymes, there is no line of defense against reactive oxygen species building up to toxic levels in red blood cells [2].
Gene and Protein
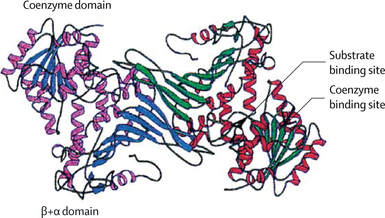
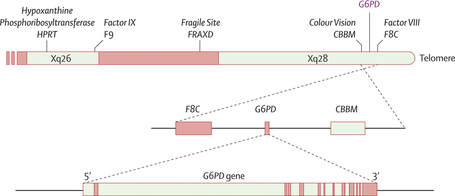
The location of the G6PD gene is Xq28 (the X chromsome). It is made up of 13 exons and 12 introns for a total of 515 amino acids. The G6PD protein functions as either a dimer or a tetramer. Each monomer of the enzyme consists of two domains, the NAD Binding Domain and the C-terminal Domain[1]. |
Figure 1. The Pentose Phosphate Pathway generates pentose sugars and NADPH from glucose.
Figure 3. Active G6PD dimer. Each subunit contains a binding domain and an active domain.
|
What is G6PD Deficiency?
|
G6PD deficiency is the most common enzymopathy in the world, with over 400 million people worldwide born with a deficient G6PD gene. Lack of functional G6PD results in insufficient NADPH production, leaving cells defenseless against toxic reactive oxygen species. The low production of NADPH can cause acute hemolytic anemia, chronic non-spherocytic hemolytic anemia, and neonatal jaundice [3]. However, the genetic deficiency is benign until triggered by an exogenous factor such as illness, antimalarials, or certain food. Over 200 different single-base-pair mutations can cause G6PD deficiency [1]. These point mutations disrupt the normal structure and function of the enzyme, or reduce the amount of the enzyme produced in cells.
|
Figure 3. The G6PD gene is located on the X-chromosome near the gene that codes for color vision.
Figure 4. G6PD catalyzes NADP+ to its reduced form, NADPH
|
Diseases Caused by G6PD Deficiency
Acute Hemolytic AnemiaRed blood cells are destroyed faster than the body can replace them. This can cause insufficient levels of oxygen to be carried throughout the body.
|
Neonatal JaundiceBilrubin is a yellow substance that builds up when red blood cells are destroyed. This occurs more often in neonates because their red blood cell turnover is so high.
|
Primaquine SensitivityPrimaquine is the only known antimalarial that targets hepatic stages of vivax malaria, preventing relapse. However, Primaquine can be fatal for those with G6PD deficiency.
|
Balancing Selection of G6PD Deficiency and Malaria
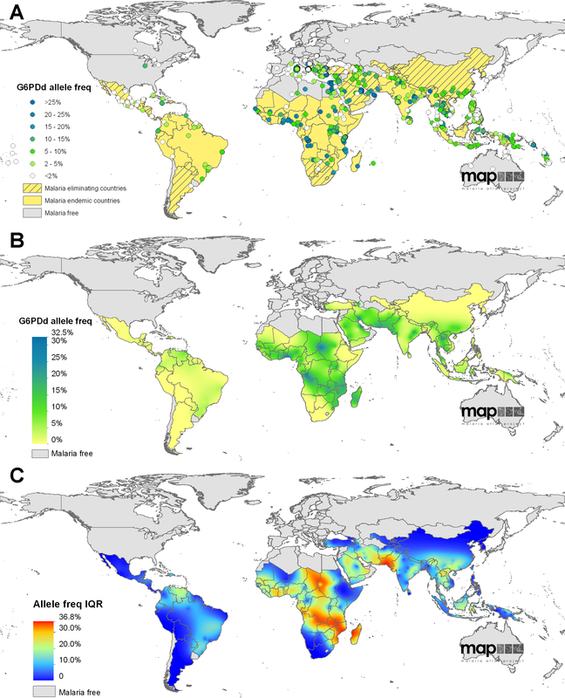
G6PD deficiency can have deleterious to fatal results. How has it persisted in populations worldwide over time? One explanation is that G6PD deficiency provides some resistance against malaria [5]. Over time, G6PD deficiency has been selected for, as red blood cells which are more susceptible to hemolysis by toxic oxidation are also less likely to be infected with the malarial parasite. As shown in the maps below, G6PD deficiency has been most strongly selected for in areas that have had high malaria endemicity in the past - Africa, Asia, and the Mediterranean region. The interaction between G6PD and malaria is an example of balancing selection; individuals with healthy red blood cells are more likely to contract malaria, but individuals with G6PD deficiency who contract malaria risk hemolytic anemia, and even death, if treated with the antimalarial Primaquine.
Figure 5. Geostatistical Map showing the overlap between malaria endemicity and G6PD deficiency
X-Linked Inheritance
Homozygous, Heterozygous, or Hemizygous
|
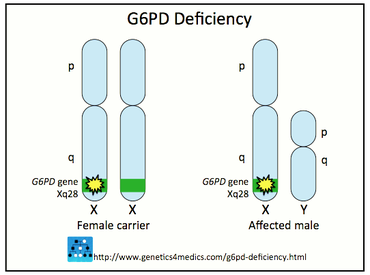
Because G6PD is located on the X-chromosome, G6PD deficiency is an "X-Linked Disease." This means that females can either be unaffected, heterozygous, or homozygous for the genetic deficiency, while males are either hemizygous or unaffected. Due to the random nature of X-inactivation in females, heterozygous females can have a wide range of the deficiency depending on which X-chromosome is inactivated in each cell. Scientists refer to this random pattern of G6PD deficiency as "erythrocyte mosacism" [5].
|
Figure 6. G6PD deficiency is X-linked. Only females can have an intermediate phenotype.
|
G6pd Deficiency Foundation, Inc.
PO Box 156
Babylon, NY 11702
631.946.7335
PO Box 156
Babylon, NY 11702
631.946.7335
References
[1] Cappellini M. (2008). "Glucose-6-phosphate dehydrogenase deficiency". The Lancet 371: 64-74
[2] Naylor C. (1996). "Glucose 6-phosphate dehydrogenase mutations causing enzyme deficiency in a model of the tertiary structure of the human enzyme". Blood 87: 2974 - 2982
[3] Luzzatto L. (2006). "Glucose 6-phosphate Dehydrogenous Deficiency: From Genotype To Phenotype". Journal of European Hematology Association 91: 1303-1306
[4] Verrelli B. (2002). "Evidence for Balancing Selection from Nucleotide Sequence Analyses of Human G6PD". American Journal of Human Genetics 71: 1112-1128
[5] Luzzato L. (1969). "Glucose-6-phosphate dehydrogenase deficient red cells: resistance to infection by malarial parasites". Science 164: 839-842
[6] Howes R. (2012). "G6PD Deficiency Prevalence and Estimates of Affected Populations in Malaria Endemic Countries: A Geostatistical Model-Based Map". PLOS Medicine online.
Figures
Figure 1. Tian W. (1998). "Importance of GLucose-6-phosphate Dehydrogenase Acitivty for Cell Growth" The Journal of Biological Chemistry 273: 10609-10617
Figure 2, 3. Cappellini M. (2008). "Glucose-6-phosphate dehydrogenase deficiency". The Lancet 371: 64-74
Figure 4. Frank J. (2005). "Diagnosis and Management of G6PD Deficiency." American Family Physician 72(7): 1277-1282.
Figure 5. Howes R. (2012). "G6PD Deficiency Prevalence and Estimates of Affected Populations in Malaria Endemic Countries: A Geostatistical Model-Based Map". PLOS Medicine online.
Figure 6. Genetics for Medics. Retrieved from http://genetics4medics.com/g6pd-deficiency.html
|
|